1. IVC Filter Manufacturers
While a variety of companies manufactured this type of product, those at the center of these allegations are C.R. Bard and Cook Medical. These manufacturers are responsible for the following products – products that have been at the center of IVC filter complication complaints.
- Recovery Filter – Bard
- G2 Filter – Bard
- G2 Express Filter – Bard
- Gunther Tulip Filter – Cook
- Celect Filter – Cook
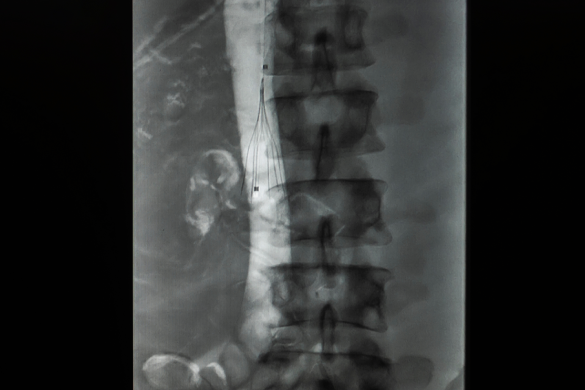
2. IVC Filter Removal / Retrieval
There are two main categories or types of IVC filters – permanent and temporary (retrievable). Those who received a retrievable filter rely on their doctors to tell them when would be an appropriate time to remove the device. To learn more about IVC filter retrieval, visit the FDA’s updated safety communication.
3. Who Might Have an IVC Filter?
Doctors can recommend IVC filter implants to those at risk for developing blood clots, such as people who recently underwent surgery, those who were recently in a serious accident, or individuals who cannot take traditional blood thinners. For example, a doctor might recommend an IVC filter to an individual who was recently in a debilitating car accident, or someone who is currently undergoing treatment for cancer.
4. Extent of Adverse Events
In August 2010, the FDA released a “risk of adverse events with long term use” safety alert regarding IVC filters. At that time, over five years ago, the FDA had received reports of:
- 921 device adverse events
- 328 of those were device migrations
- 146 of those were embolizations
- 70 of those involved IVC perforation
- 56 involved filter fracturing
5. Device Migrations
The most commonly reported adverse event associated with IVC filter is device migration, which describes an event where the filter moves from its original place in the inferior vena cava to another region – even the heart or lungs.
According to a study published in the National Center for Biotechnology Information (NCBI), “newer retrievable filters made of nitinol, phynox and elgioly have a significantly higher percentage of filter migration into the right ventricle as compared to the old stainless steel and titanium-based Greenfield filters. Similarly, there were also higher percentages of complications and mortality associated with the newer retrievable filters migrating to the right ventricle.”
6. IVC Filter Embolization
Another study posted in the NCBI concluded that Bard Recovery and Bard G2 filters in particular had a high prevalence of life-threatening embolization. Embolization is another way to describe detachment of device components (ex: prongs fracturing off the device).
7. IVC Filter Perforation
Around 7% of all adverse events reported in connection with the IVC filter involved perforation. Perforation is often listed in connection with device migration or embolization, because it involves all or part of the device migrating from the inferior vena cava and perforating a nearby organ.
8. IVC Filter Fractures
IVC filters have reportedly fractured in some instances, sending pieces of the filter into patients’ hearts, lungs, and other areas. IVC filter fractures can cause permanent damage to organs, and can be life threatening.
9. Unsuccessful Removable IVC Filters
A study published in the Journal of the American Medical Association stated that out of 680 patients who were implanted with retrievable IVC filters, only 58 were able to be removed. Some doctors reported unsuccessful removal of retrievable IVC filters due to device embedding, device protrusion through the blood vessel, device migration, and blood clot inside the filter.
10. Legal Recourse for IVC Filter Injuries
If you or someone you love was implanted with the IVC filter and experienced complications, such as those described in this article, we encourage you to get in touch with us today to learn your legal rights and options. Bailey Cowan Heckaman PLLC PLLC is a national trial law firm located in Houston. We have a reputation for producing winning results for the seriously injured, so get in touch with us today for a free review to discuss your IVC filter case and what happened to you.